The Problem
- Emerging variants threatened to erode vaccine protection, creating need for real-time effectiveness data.
- Traditional clinical studies lagged in measuring vaccine performance across diverse regions and variant strains.
- Policymakers lacked timely insight on whether vaccines remained protective, risking delayed booster responses.
The Solution
- Leveraged a global Facebook user survey to gather self-reported vaccination status and COVID outcomes daily across 100+ countries.
- Analyzed millions of responses to compare COVID-like illness rates in vaccinated vs unvaccinated populations over variant waves.
- Used statistical models to estimate infection risk reduction by vaccine status for each variant, adjusting for demographics.
Architecture Overview
- Data Integration: Aggregated daily survey responses and aligned them with variant prevalence timelines from genomic surveillance.
- Variant Segmentation: Tagged survey data by time and region to correlate infection rates with dominant variants (e.g. Delta, Omicron).
- Effectiveness Modeling: Employed statistical and ML models to compute odds ratios of infection for vaccinated vs unvaccinated groups per variant.
- Dashboard & Visualization: Built a dashboard to visualize vaccine performance trends over time and across regions as variants arose.
- Quality Control: Weighted survey data for representation and validated model outputs against external case and hospitalization data.
Results and Impacts
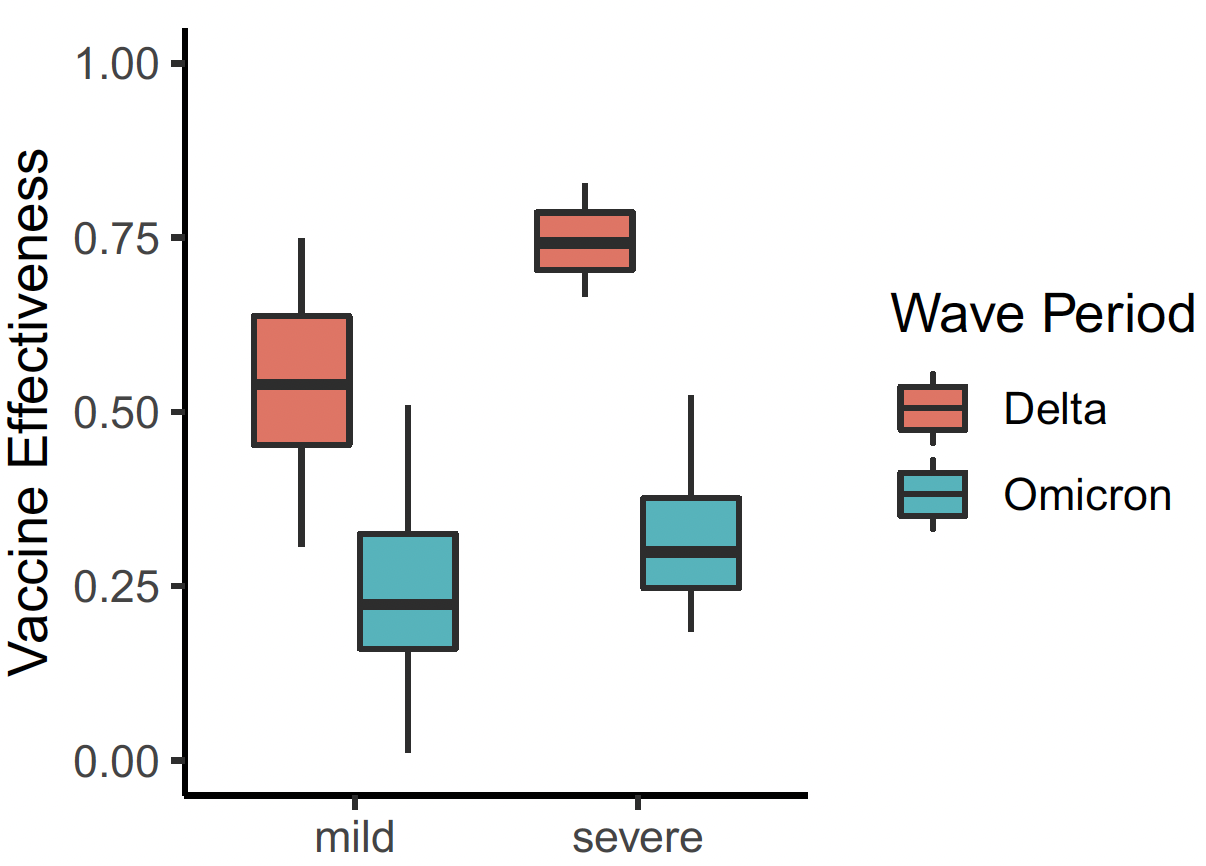
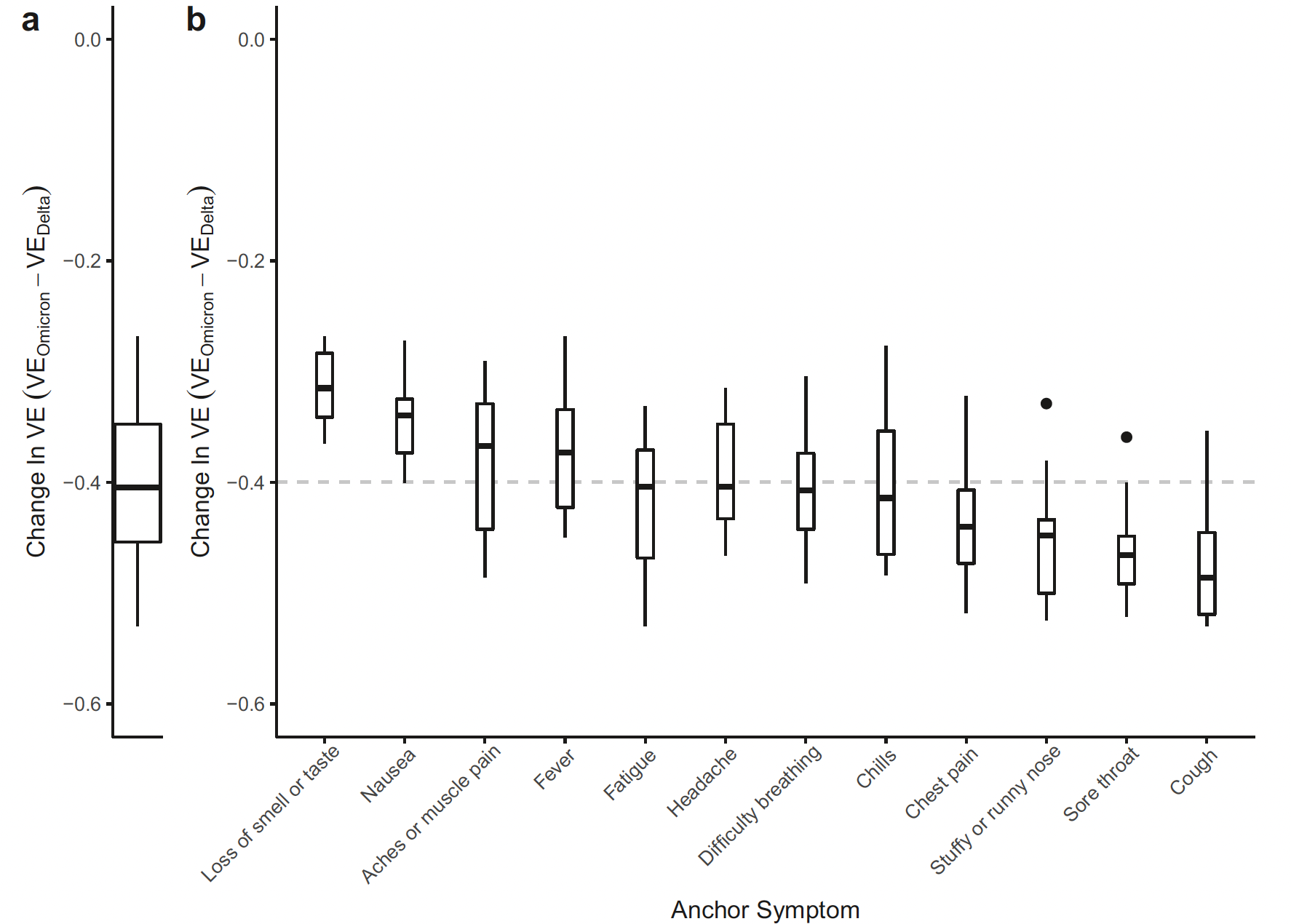
- Demonstrated high vaccine effectiveness (~85%) against early COVID strains, with moderate declines against Delta and further reduction against Omicron.
- Real-time analysis flagged waning vaccine protection with new variants, supporting timely public health decisions on booster recommendations.
- Showed that participatory survey data can rapidly evaluate vaccine performance globally, influencing policy in areas lacking formal studies.
Skills and Tools Used
| Technique/Skill | Tools/Implementation |
|---|---|
| Data Analysis | Large-scale survey data processing (Python, R) |
| Epidemiological Modeling | Vaccine effectiveness calculations (odds ratios, risk reduction) |
| Data Integration | Merged survey outcomes with variant prevalence data |
| Cloud & Collaboration | Scalable cloud pipeline; coordinated analysis across institutions |
Cross-Project Capabilities
- Rapid Public Health Analysis: Ability to quickly analyze global data for urgent health decisions (e.g., pandemic response).
- Adaptive Modeling: Experience adjusting models for evolving conditions (new variants) transferable to other dynamic scenarios.
- Global Survey Expertise: Strengthened skills in using large participatory datasets to complement traditional surveillance in various domains.
Published Papers/Tools
- Study results published in Nature Communications Medicine (2024), detailing vaccine efficacy trends across variants. Paper
- Findings shared with global health authorities, providing data-driven guidance for booster policies and pandemic planning.